
Circadian Rhythms:
Almost every aspect of plant and animal biology displays 24 hours rhythms as the result of the rotation of our planet on its axis and the corresponding profound changes in the environment which constitute the day-night cycle. Many of these rhythms persist under constant conditions, although in this artificial situation their period lengths are usually a little longer or shorter than 24 hours. For this reason, they are termed circadian (Circa – diem; approximately one day) rhythms. The mechanism that directs circadian rhythms is the Circadian Clock1.
The Circadian Timing System:
Central to the generation of circadian rhythms is a self-sustaining, endogenous cellular clock mechanism which oscillates even under artificial constant conditions. However, to ensure that this approximately 24 hours Clock mechanism remains synchronized with the 24 hours environmental cycle, it is essential that it is reset (“entrained”) on a daily basis by environmental factors such as light, temperature, food or social behaviour which are indicative of the passage of the day-night cycle (so-called “zeitgebers” or time-givers) 2. The detection and relaying of zeitgeber information from the environment to the circadian clock occurs via clock “input” pathways. In turn, the clock directs circadian rhythmicity in most biological systems via an array of clock “output” pathways. The combination of the clock, clock input and clock output pathways is termed the Circadian Timing System1 Figure 1. At a “system level”, the circadian timing system can be considered as a three-part mechanism: 1) The central clock which generates circadian rhythms, even independently of the environment. 2) An input pathway whereby signals indicative of the time of day such as light, temperature and food (so-called Zeitgebers) set the phase of the clock and ensure it remains synchronized with the environment and finally 3) output pathways whereby the clock conveys timing information to various aspects of behaviour and physiology.
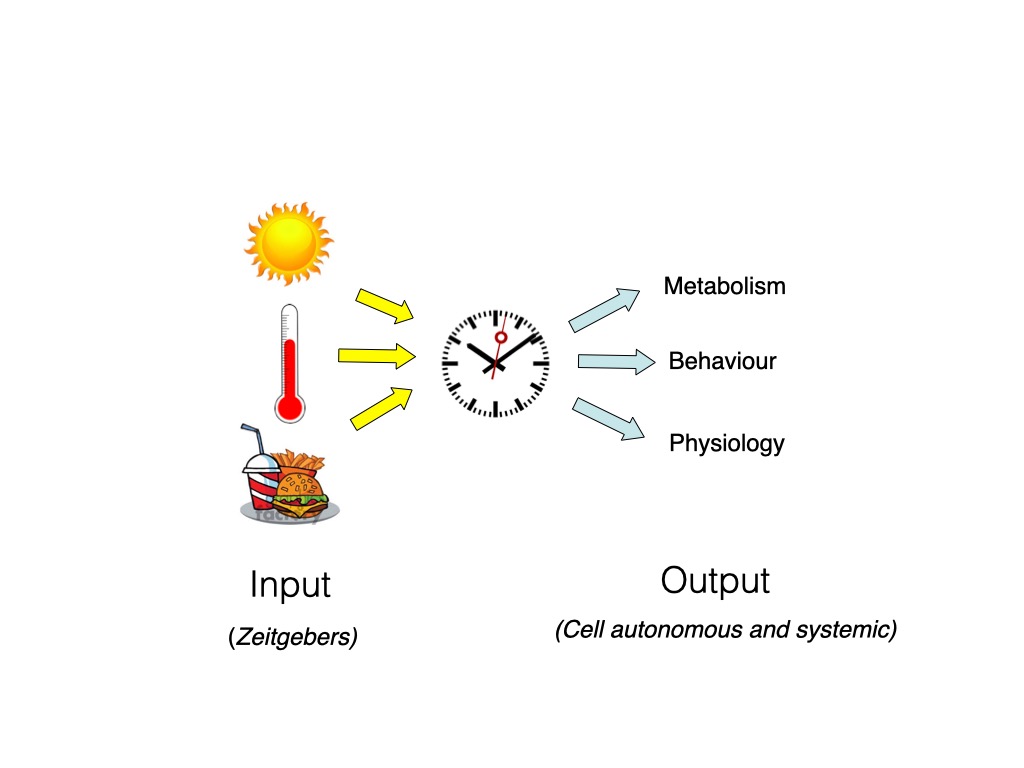
In vertebrates, specialized so-called “central pacemakers” are located in the suprachiasmatic nucleus (SCN) of the hypothalamus and the retina. In lower vertebrates, the pineal gland also appears to contain such a pacemaker. The integrity of these structures is essential for normal clock function in an organism. However, circadian clocks are not restricted to specialized cell types or tissues. They are actually encountered in most cell types and tissues. At the organismal level, the circadian timing system has been likened to an orchestra where central pacemakers such as the SCN serve as conductors, which ensure that the multiple tissue and cell clocks, the so-called peripheral clocks, remain synchronized with each other 3,4.
In mammals, the SCN clock is entrained by light, exclusively via photoreceptors in the retina: intrinsically photosensitive retinal ganglion cells (ipRGCs) which express the non-visual opsin, melanopsin. These cells are distinct from the rod and cone visual photoreceptor cells. In contrast, in lower vertebrates such as fish, all central and peripheral clocks are directly light entrainable via widely expressed photoreceptors. These light sensitive peripheral clocks function even in cell cultures5,6 .
The clock output pathways which convey timing information from the circadian clock to the biological systems it regulates appear to be complex. They range from the direct regulation of clock-controlled gene (CCG) expression by clock proteins, clock controlled humoral signals (such as rhythmic hormone levels and daily changes in body temperature) to circadian rhythms of neuronal signals 4,7,8.
Clock Genes and the Circadian Clock Mechanism:Large scale genetic analysis of the clock in a wide range of genetic model organisms (cyanobacteria, fungi Neurospora, plants (Arabidopsis), mice and humans lead to the identification of the components of the clock mechanism: so-called clock genes and clock proteins9. Many clock gene products function as transcription factors which ultimately activate or repress their own expression. They therefore constitute what are termed autoregulatory transcription-translation feedback loops. Changes in the subcellular localization and posttranslational modifications of clock proteins as well as delays between transcription and translation of clock genes seem to be crucial for conferring the relatively long time (approximately 24 hours) it takes the clock mechanism to complete one cycle. Furthermore, the mechanism includes multiple, interlocking feedback loops, much like the many cogs of a real clock, which work to stabilize the circadian clock mechanism10,11 .
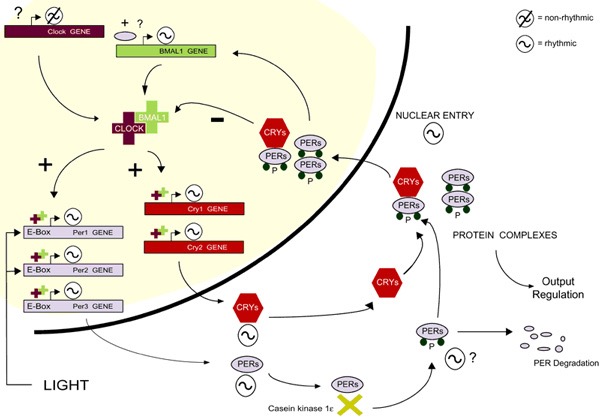
As well as regulating the expression of clock genes, clock proteins can also serve as transcriptional regulators of CCGs and therefore constitute an important clock output pathway8 . The acute induction of the expression of a subset of clock genes, (a subset of per and cry genes in vertebrates) in response to exposure to particular zeitgebers, appears to serve as a conserved mechanism for synchronizing the phase of the clock rhythm relative to the day night cycle. Light represents the best studied zeitgeber and the photoreceptors that specifically detect light for entraining the clock are commonly termed circadian photoreceptors12. In mammals, melanopsin which is a non-visual opsin expressed exclusively in ipRGC cells within the retina, serves as the principal circadian photoreceptor. A neuronal signal originating from the detection of light by ipRGC melanopsin is relayed to adjust the phase of the SCN clock indirectly via the Retino-Hypothalamic Tract (RHT). The SCN clock in turn adjusts the peripheral clocks via a complex combination of humoral signals13 .
What have we learned from studying the circadian clock in zebrafish?
Although historically the zebrafish represent one of the most important model organisms for studying embryonic development in vertebrates14, it has also emerged as a very attractive model for studying the circadian timing system. As well as providing a powerful collection of genetic tools for studying clock mechanisms in vivo, fish possess directly light-entrainable peripheral clocks, even in fish-derived cell lines. Therefore, this has made the zebrafish particularly useful for studying the pathways conveying light information to the circadian clock15.

The precise nature of the widely expressed circadian photoreceptors in fish remains unclear. Teleosts possess an unusually large repertoire of opsins (43 identified to date in the zebrafish genome), with the majority being expressed outside of dedicated photoreceptive tissues such as the retina and pineal gland. These include teleost-specific opsins such as teleost multiple tissue (TMT) opsin as well as homologs of melanopsin, the principle non-visual circadian photoreceptor in mammals16. The striking diversity and expression pattern of opsins in fish tissues is consistent with them serving as peripheral photoreceptors. Furthermore, other non-opsin-based photoreceptor systems, have also been implicated in peripheral photoreception in zebrafish, including flavin-containing oxidases which generate ROS species upon exposure to light17,18 and also certain cryptochrome homologs which can serve as blue light photoreceptors in plants and drosophila19.
Sunlight-regulated gene expression:
We have focused on the establishment of cell cultures from embryonic zebrafish embryos, as well as from adult caudal fins. Combined with the transfection of these cells with bioluminescent promoter reporter constructs, we have been able to study the mechanisms whereby sunlight exposure regulates gene expression20,21.

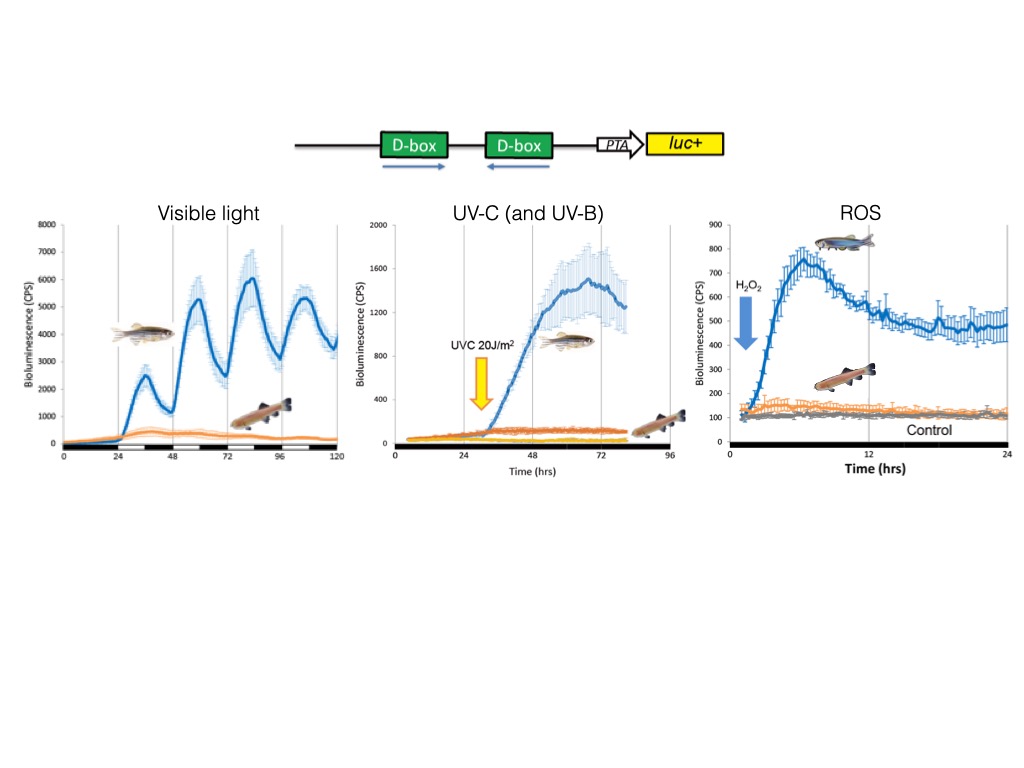
This approach has allowed us to explore how light, temperature, ROS or UV regulate the expression of the per1b21, cry1a22 and per223 clock genes as well as DNA repair genes24 that in zebrafish are also transcriptionally activated by visible light. We have pinpointed a light, ROS and UV responsive enhancer element, the D-box element17,22–24 which operates alone or in combination with other enhancers such as the E-box or E2F sites23.
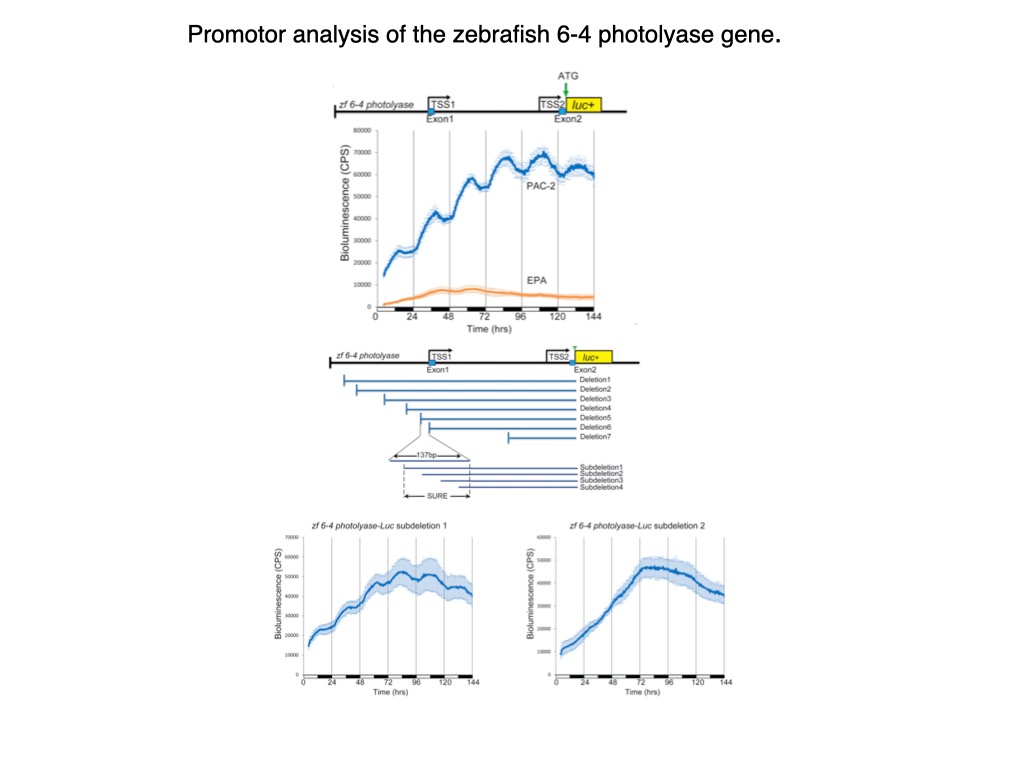
.
In contrast to this situation in fish, in mammals the D-box serves as an element of the circadian clock output pathway suggesting that D-box function has changed significantly during vertebrate evolution23,25. Using pharmacological approaches, we have demonstrated that exposure to short wavelengths of visible light in zebrafish cells triggers increases in ROS levels via NADPH oxidase activity. Elevated ROS activates the JNK and p38 MAP kinases that in turn, induces clock gene expression via D-box elements17.
Cell cycle and the clock:
One of the main cellular processes regulated by the circadian clock that we have studied in our laboratory is cell cycle progression. The timing of cell proliferation is a key factor contributing to the regulation of normal growth. In turn, circadian clock disruption has been linked to an increased incidence of cancer26,27 In animals subjected to prolonged jet lag or constant light exposure as well as in night shift workers there is an increased risk of certain types of cancer. In addition, studies in rodents where the circadian clock has been genetically disrupted, revealed an increase in tumorigenesis after inoculation with tumor cells. Moreover, because cancer patients commonly show circadian clock disruption, the degree of circadian clock synchronization has become a prognostic tool for breast cancer patients28.
Daily rhythms of cell cycle progression have been documented in a wide range of organisms. Our group demonstrated that exposure of zebrafish larvae or adults to light-dark cycles causes a range of different cell types to enter S-phase predominantly at the end of the light period29,30.
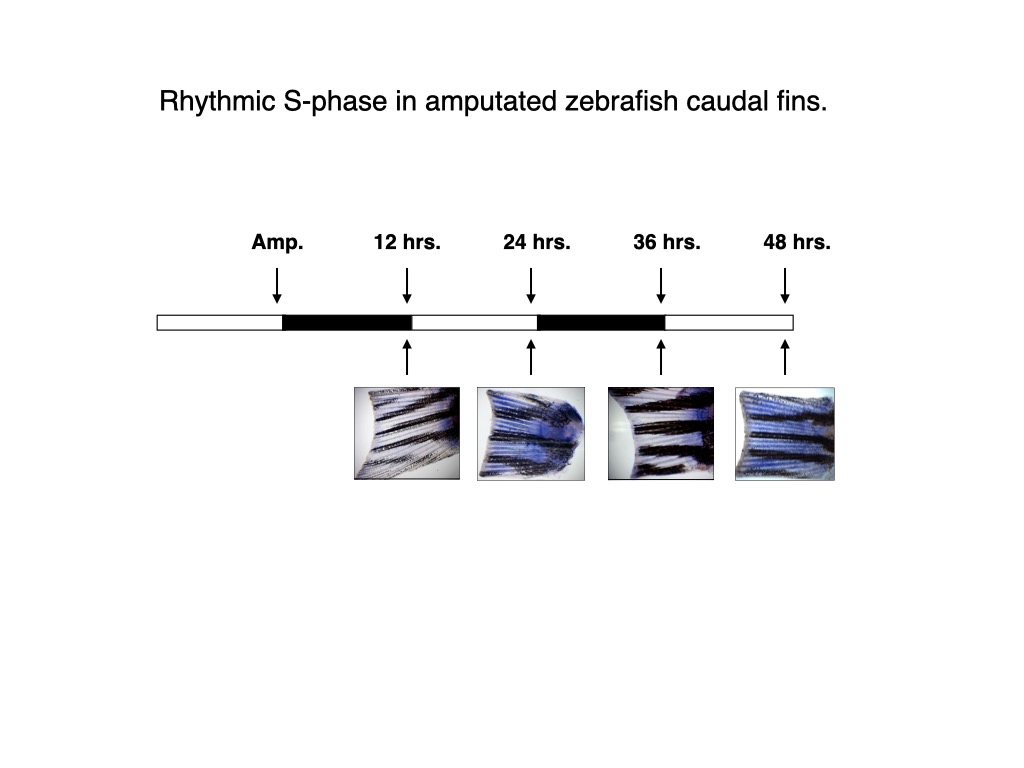
.
This rhythm is conserved for several days after transfer of the larvae in constant darkness and is also conserved in an in vitro cell culture system pointing to cell autonomous circadian clock regulation. Interestingly, constant levels of glucocorticoids are also required for a sustained circadian cell cycle rhythmicity31,32. In addition, we encountered rhythmic mRNA expression for the regulator of the G1/S and G2/ M cell cycle transition zf p21, zf cyclinA2, zf cyclin B1and the kinase zf wee130.
We have also implicated the Y box binding 1 (YB-1) oncoprotein as one of the links between the circadian clock and the control of cell cycle33. Specifically, using immunofluorescence and western blot analysis, we have demonstrated in vivoand in vitro that the nuclear concentration of YB-1 protein in zebrafish undergoes a daily oscillation with a period of circa 24 hours.
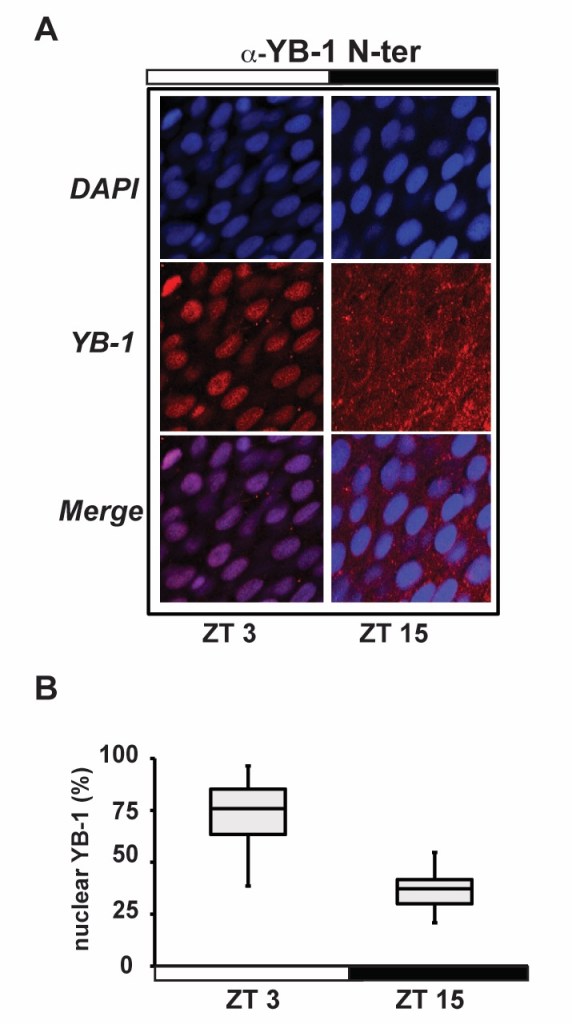
This oscillation results in accumulation of YB-1 in the nucleus during the first hours of the light phase with a peak at 3- hours after lights ON (zeitgeber time, ZT 3-6) and a reduction to a minimum level around the beginning of the dark phase (ZT 12-15). This oscillation in YB-1 expression has been associated with changes in protein SUMOylation and with a transcriptional inhibition of the cell cycle regulatory gene zf cyclin A2.
Studying other fish species:Teleosts represent by far the largest and most diverse group of vertebrates, with something like 25.000 – 30.000 known species. They occupy a huge diversity of environments which include extreme habitats. This makes fish eminently well suited for studying the influence of the environment in shaping evolution of the circadian timing system5. A good example is the perpetually dark cave environment where certain teleost species have evolved in complete isolation from the day night cycle. Blind cavefish represent a group of 100 – 200 unrelated species distributed worldwide which inhabit subterranean bodies of water and share a set of striking physiological and anatomical adaptations for life under complete darkness. These so-called troglomorphisms include complete eye loss and absence of body pigmentation. However, blind cavefish are also attractive models for studying how the absence of the day-night cycle shapes the evolution of the circadian clock and other sunlight related functions such as DNA repair. To date, the circadian timing system has been studied intensively in two species of cavefish, namely Phreatichthys andruzzii and Astyanax mexicanus5. In our laboratory, we have focused on P. andruzzii that is a Somalian cavefish which exhibits an extreme troglomorphic phenotype.

This fish inhabits layers of water contained within limestone rock formations deep beneath the Somalian desert. The only contact between the water layers where P.andruzzii lives and the surface are small, deep natural wells in the desert. Based on the geology of the region, it is predicted that these animals have been isolated from the surface for over 3 million years as a result of desertification in that region of Africa. In our laboratory, comparing molecular and behavioural data of P. andruzzii to a surface-dwelling cyprinid, the zebrafish, has shed new light on the evolution of the circadian clock5,17,24,34,35.
Specifically, artificial exposure of P. andruzzii to Light/Dark cycles resulted in an arrhythmic pattern of both locomotor activity and clock gene expression in the brain and peripheral tissues, as well as in primary cell lines derived from adult fins and embryos34.
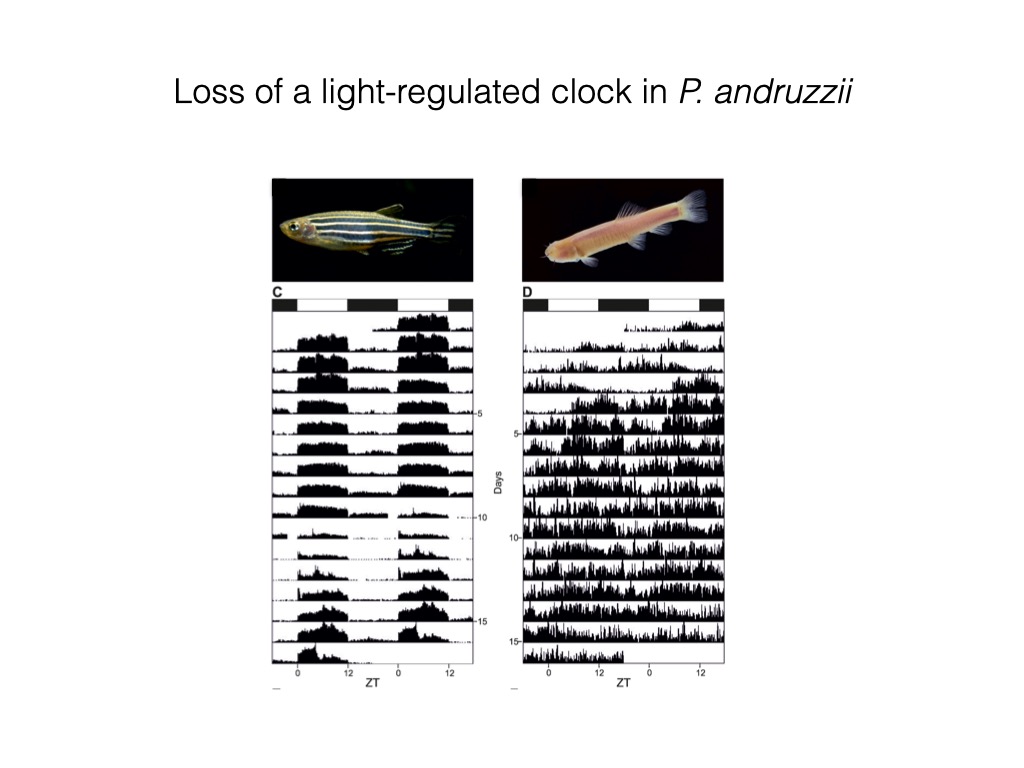
We thus demonstrated that this cavefish species has lost the capacity to entrain the circadian clock by light, in vivo and in vitro. However, these animals are still able to entrain their circadian clocks in response to regular feeding time34.
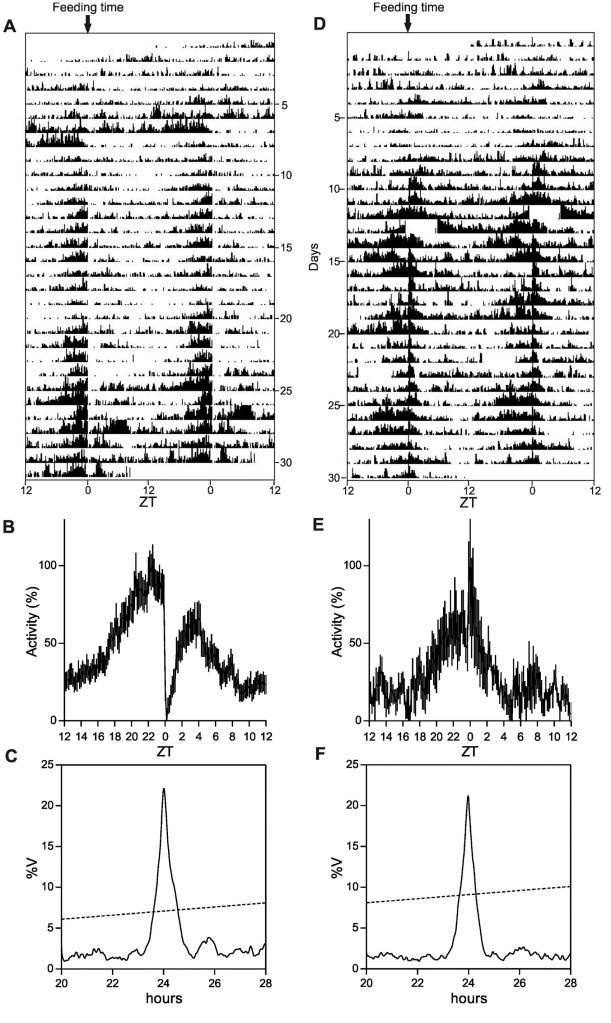
.
Therefore, P. andruzzii still has a functional circadian clock, but this clock is completely “blind”. We revealed that the mutation of two nonvisual opsin genes was at least in part responsible of this loss of light entrainment34,35. Furthermore, we have identified a loss of function of the regulatory pathway whereby visible light, ROS and UV activate expression of the three photolyase genes (6-4 phr, DASHphr and CPDphr)24. The photolyases perform photoreactivation DNA repair, whereby visible light is harnessed to catalyse the repair of UV damaged DNA. We revealed that the cavefish P. andruzzii has lost photoreactivation repair primarily due to a loss of D-box enhancer function in the promoters of the photolyase genes
Curiously, photoreactivation is highly conserved throughout eukaryotes and prokaryotes with the notable exception of placental mammals and now, the blind cavefish, P. andruzzii. It is tempting to speculate that the loss of photoreactivation in this cavefish is related to the loss of UV damage in the cave environment as well as the absence of visible light required to drive this reaction. In turn, we might speculate that the loss of this DNA repair mechanism in the ancestors of the placental mammals might have resulted from a period of evolution in a predominantly dark, subterranean environment. Interestingly, this notion would tend to support the “nocturnal bottleneck theory” which predicts that the early mammals were burrowing, predominantly nocturnal animals in order to survive predation by the dominant carnivorous dinosaurs of that era36.
References.
1. Pittendrigh, C. S. Temporal organization: reflections of a Darwinian clock-watcher. Annual review of physiology 55, 16–54 (1993).
2. Roenneberg, T., Daan, S. & Merrow, M. The art of entrainment. Journal of Biological Rhythms 18, 183–194 (2003).
3. Schibler, U. & Sassone-Corsi, P. A web of circadian pacemakers. Cell 111, 919–922 (2002).
4. Schibler, U. et al. Clock-Talk: Interactions between Central and Peripheral Circadian Oscillators in Mammals. Cold Spring Harbor symposia on quantitative biology 027490 (2015) doi:10.1101/sqb.2015.80.027490.
5. Foulkes, N. S., Whitmore, D., Vallone, D. & Bertolucci, C. Studying the Evolution of the Vertebrate Circadian Clock. in vol. 95 1–30 (Elsevier, 2016).
6. Menaker, M., Moreira, L. F. & Tosini, G. Evolution of Circadian Organization in Vertebrates. Braz J Med Biol Res 30, 305–313 (1997).
7. Morf, J. & Schibler, U. Body temperature cycles: gatekeepers of circadian clocks. Cell cycle (Georgetown, Tex) 12, 539–540 (2013).
8. Hughes, M. E. et al. Harmonics of Circadian Gene Transcription in Mammals. PLoS genetics 5, e1000442-12 (2009).
9. Lowrey, P. L. & Takahashi, J. S. Genetics of circadian rhythms in Mammalian model organisms. Advances in genetics74, 175–230 (2011).
10. Vanselow, K. & Kramer, A. Role of phosphorylation in the mammalian circadian clock. Cold Spring Harbor symposia on quantitative biology 72, 167–176 (2007).
11. Reppert, S. M. & Weaver, D. R. Molecular analysis of mammalian circadian rhythms. Annual review of physiology63, 647–676 (2001).
12. Hughes, S., Jagannath, A., Hankins, M. W., Foster, R. G. & Peirson, S. N. Photic regulation of clock systems. Methods in enzymology 552, 125–143 (2015).
13. Doyle, S. & Menaker, M. Circadian photoreception in vertebrates. Cold Spring Harbor symposia on quantitative biology 72, 499–508 (2007).
14. Mullins, M. C., Hammerschmidt, M., Haffter, P. & Nusslein-Volhard, C. Large-scale mutagenesis in the zebrafish: in search of genes controlling development in a vertebrate. Current Biology 4, 189–202 (1994).
15. Steindal, I. F. & Whitmore, D. Circadian Clocks in Fish—What Have We Learned so far? Biology 8, 17 (2019).
16. Davies, W. I. L. et al. An extended family of novel vertebrate photopigments is widely expressed and displays a diversity of function. Genome research 25, 1666–1679 (2015).
17. Pagano, C. et al. Evolution shapes the responsiveness of the D-box enhancer element to light and reactive oxygen species in vertebrates. Scientific Reports 8, 1–17 (2018).
18. Hirayama, J., Cho, S. & Sassone-Corsi, P. Circadian control by the reduction/oxidation pathway: Catalase represses light-dependent clock gene expression in the zebrafish. Proceedings of the National Academy of Sciences of the United States of America 104, 15747–15752 (2007).
19. Sancar, A. Structure and function of DNA photolyase and cryptochrome blue-light photoreceptors. Chemical reviews 103, 2203–2237 (2003).
20. Vallone, D., Santoriello, C., Gondi, S. B. & Foulkes, N. S. Basic protocols for zebrafish cell lines: maintenance and transfection. Methods in molecular biology (Clifton, N.J.) 362, 429–441 (2007).
21. Vallone, D., Gondi, S. B., Whitmore, D. & Foulkes, N. S. E-box function in a period gene repressed by light. Proceedings of the National Academy of Sciences 101, 4106–4111 (2004).
22. Mracek, P. et al. Regulation of per and cry Genes Reveals a Central Role for the D-Box Enhancer in Light-Dependent Gene Expression. PLoS ONE 7, e51278 (2012).
23. Vatine, G. et al. Light Directs Zebrafish period2 Expression via Conserved D and E Boxes. PLoS Biology 7, e1000223 (2009).
24. Zhao, H. et al. Modulation of DNA Repair Systems in Blind Cavefish during Evolution in Constant Darkness. Current Biology 28, 3229-3243.e4 (2018).
25. Gachon, F. Physiological function of PARbZip circadian clock‐controlled transcription factors. 39, 562–571 (2009).
26. Shafi, A. A. & Knudsen, K. E. Cancer and the Circadian Clock. Cancer Res 79, 3806–3814 (2019).
27. Hernández-Rosas, F., López-Rosas, C. A. & Saavedra-Vélez, M. V. Disruption of the Molecular Circadian Clock and Cancer: An Epigenetic Link. Biochem Genet 58, 189–209 (2020).
28. Reszka, E. & Przybek, M. Chapter 3: Circadian Genes in Breast Cancer. Adv Clin Chem 75, 53–70 (2016).
29. Dekens, M. P. S. et al. Light regulates the cell cycle in zebrafish. Current biology : CB 13, 2051–2057 (2003).
30. Idda, M. L. et al. Circadian Timing of Injury-Induced Cell Proliferation in Zebrafish. PLoS ONE 7, e34203 (2012).
31. Dickmeis, T. et al. Glucocorticoids play a key role in circadian cell cycle rhythms. PLoS biology 5, e78 (2007).
32. Dickmeis, T. & Foulkes, N. S. Glucocorticoids and circadian clock control of cell proliferation: at the interface between three dynamic systems. Molecular and cellular endocrinology 331, 11–22 (2011).
33. Pagano, C. et al. The tumor-associated YB-1 protein: new player in the circadian control of cell proliferation. Oncotarget 5, (2016).
34. Cavallari, N. et al. A Blind Circadian Clock in Cavefish Reveals that Opsins Mediate Peripheral Clock Photoreception. PLoS Biology 9, e1001142 (2011).
35. Calderoni, L. et al. Relaxed selective constraints drove functional modifications in peripheral photoreception of the cavefish P. andruzzii and provide insight into the time of cave colonization. Heredity 117, 383–392 (2016).
36. Gerkema, M. P., Davies, W. I. L., Foster, R. G., Menaker, M. & Hut, R. A. The nocturnal bottleneck and the evolution of activity patterns in mammals. Proceedings. Biological sciences / The Royal Society 280, 20130508–20130508 (2013).